|
Some reactions release energy in the form of heat, light, or sound, while others absorb energy from the surroundings. Energy Changes: Chemical reactions involve the exchange or transformation of energy.This rearrangement of atoms leads to the formation of different substances. Breaking and Forming of Bonds: During a chemical change, the bonds between atoms in the reactant molecules are broken, and new bonds are formed to create the product molecules.Reactants are the initial substances that transform, while products are the resulting substances formed after the reaction. Reactants and Products: Chemical changes involve the conversion of reactants into products.Read More: Difference Between Physical and Chemical Changes Organic, inorganic, and biological changes are the three different types of chemical alterations.It generates heat, endothermic reactions may require heat to continue.While certain processes are known as exothermic reactions. Chemical reactions are what are known as these processes, and they are often irreversible, barring other chemical reactions.Chemical modifications result in the creation of new products. 
The transformation of one substance into another through the dissolution of existing bonds and the formation of new ones is known as a chemical change.The building and breaking of chemical bonds between atoms is a component of chemical changes, commonly referred to as chemical reactions.The phrase "chemical change" describes how one or more compounds are transformed into new substances with new attributes. Photosynthesis is also a chemical change as carbohydrate is formed as the product of the reaction between carbon di oxide an water.Examples of chemical change are The formation of curd from milk through fermentation, Rusting of iron as a form of corrosion, Acid-base titration, Burning of wood, candles, and paper, Cooking up food, Baking cakes and cookies, Digestion of food, Metal electroplating, Ripening and rotting of fruits, etc.The neutralization reaction between an acid and base is also a chemical change.This happens because of the reaction occuring between an enzyme tyrosinase and oxygen in the air. The slice appears brown on color which changes the taste of the apple slice. We have noticed the change in color if the apple is cut with the knife after some time. Spoilage of food is a chemical change.When this magnesium oxide is reacted with water it gives Magnesium hydroxide as the end product. Equation supporting this reaction has been shown below. Burning of magnesium ribbon is accompanied with a brilliant white light, leaving powdery ash behind.If this lime water turns milky it means the gas evolved is carbon dioxide. If we want to confirm it whether the gas is carbon dioxide or not, pass the gas through the lime water and observe the solution.When a pinch of baking soda is added to vinegar, production of a hissing sound is accompanied with the formation of the bubbles of gas.A new substance is formed as the end product of the reaction. A chemical reaction may also be accompanied by a change in color, production of sound, formation of gas etc.Ĥ. A chemical change also involves a chemical reaction. It can be represented by a chemical equation.ģ. In a chemical change exchange of energy takes place.Ģ. For example- Souring of milk, curding of milk, burning of curd, rusting of iron etc. In chemical change the properties of the substance changes. We consider chemical changes as irreversible changes. Grade 7 | Science | Chemical changes, Physical and Chemical changes, Olympiad, CBSE, ICSE, Maths Olympiad, Science Olympiad, English Olympiad Chemical changes A change where the original substance changes into a new substance are called the chemical change. Grade 7, Science Olympiad (CBSE) - Chemical changes  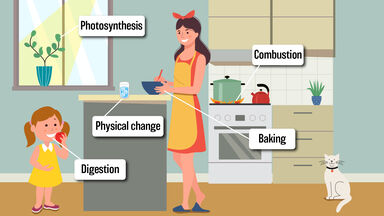
Transportation in plants (water and food).Natural resources and their conservation.Role of the meteorological department of a country.Weather, Climate and adaptation of animals to climate.Hazards and safety measures of electricity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
